Hence its co-ordination number is 6.Įight unit cells share each atom at the corner. In simple cubic structure each atom consists of 6 equidistant nearest neighbours. If the number of atoms per unit cell are n and if V a is the volume of atoms in the unit cell and V is the volume of the unit cell then,Ĭalculation of Atomic packing factor for different crystal systems 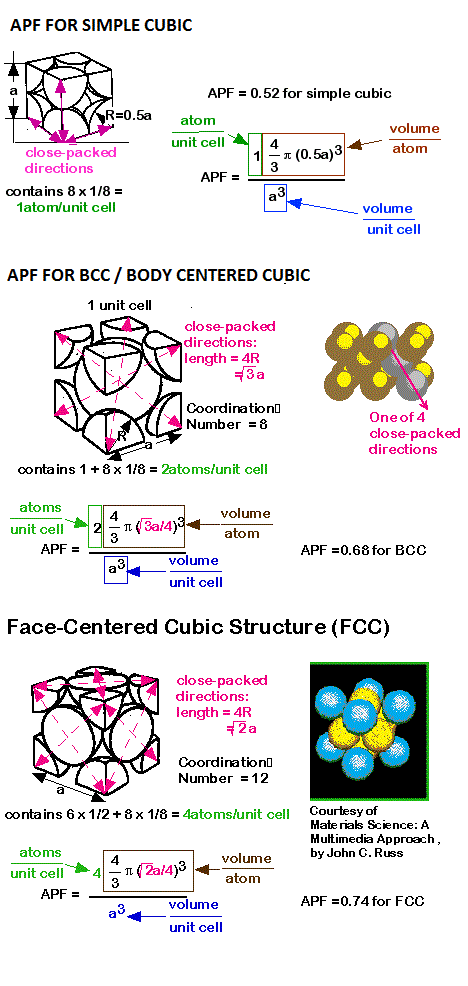
It is defined as the ratio of volume occupied by atoms in unit cell to the volume of the unit cell. It is the fraction of space occupied by atoms in a unit cell. (3) Atomic packing factor (APF) or Packing fraction: It is the distance between two nearest neighbors in a crystal structure. It is the number of equidistant nearest neighbors that an atom has in a crystal structure. The atomic packing factor of the diamond cubic structure ( the proportion of space that would be filled by spheres that are centered on the vertices of the structure and are as large as possible without overlapping ) is | 16 } } H " 0.34, significantly smaller ( indicating a less dense structure ) than the packing factors for the face-centered and body-centered cubic lattices.Coordination number and Atomic packing factor Definition When cooled quickly, the chains tightly align, giving PFO a close packing factor, though because of the high complexity of the chains, this sometimes gets messy and creates the amorphous state.Īssuming one atom per lattice point, in a primitive cubic lattice with cube side length " a ", the sphere radius would be and the atomic packing factor turns out to be about 0.524 ( which is quite low ). The atomic packing factor runs around 68-74 % in a crystal, and might be a bit worse with an amorphous solid, let's say 50 %. The coordination number of atoms in hcp and fcc structures is 12 and its atomic packing factor ( APF ) is the number mentioned above, 0.74. 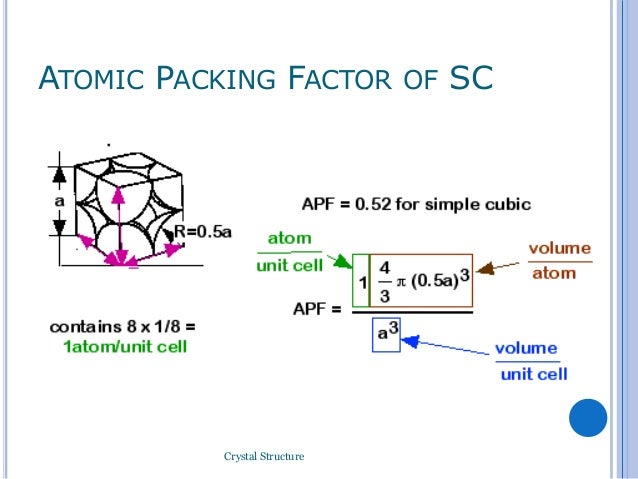
This is done by treating each cell as a single point and using spatial statistics such as the Effective Radius, Packing Factor and Regularity Index. Zincblende structures have higher packing factors than 0.34 depending on the relative sizes of their two component atoms. The atomic packing factor is the proportion of space filled by these spheres. One important characteristic of a crystalline structure is its atomic packing factor. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
March 2023
Categories |
 RSS Feed
RSS Feed
